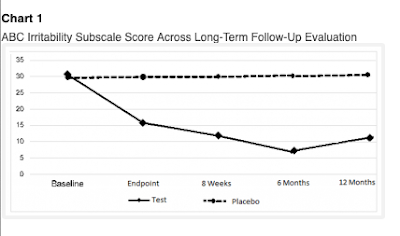
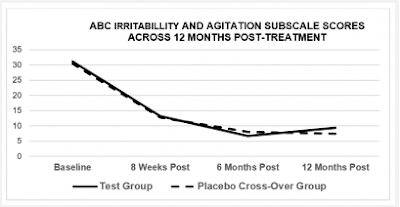
In my last blogpost, I looked at a study that claimed continuing improvements of symptoms of autism after eight 5-minute sessions where a low-level laser was pointed at the head. The data were so extreme that I became interested in the company, Erchonia, who sponsored the study and in Regulatory Insight, Inc, whose statistician failed to notice anything odd. In exploring Erchonia's research corpus, I found that they have investigated the use of their low-laser products for a remarkable range of conditions. A search of clinicaltrials.com with the keyword Erchonia produced 47 records, describing studies of pain (chronic back pain, post-surgical pain, and foot pain), body contouring (circumference reduction, cellulite treatment), sensorineural hearing loss, Alzheimer's disease, hair loss, acne and toenail fungus. After excluding the trials on autism described in my previous post, fourteen of the records described randomised controlled trials in which an active laser was compared with a placebo device that looked the same, with both patient and researcher being kept in the dark about which device was which until the data were analysed. As with the autism study, the research designs for these RCTs specified on clinicaltrials.com looked strong, with statistician Elvira Cawthon from Regulatory Insight involved in data analysis.
As shown in Figure 1, where results are reported for RCTs, they have been spectacular in virtually all cases. The raw data are mostly not available, and in general the plotted data look less extreme than in the autism trial covered in last week's post, but nonetheless, the pattern is a consistent one, where over half the active group meet the cutoff for improvement, whereas less than half (typically 25% or less) of the placebo group do so.
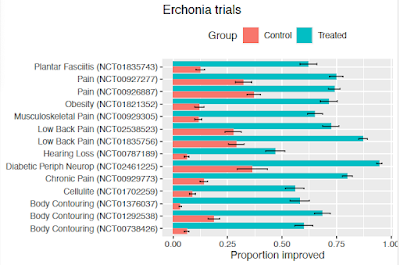 |
| FIGURE 1: Proportions in active treated group vs placebo group meeting preregistered criterion for improvement (Error bars show SE)* |
I looked for results from mainstream science
against which to benchmark the Erchonia findings. I found a big review
of behavioural and pharmaceutical interventions for obesity by the US
Agency for Healthcare Research and Quality (LeBlanc et al, 2018).
Figures 7 and 13 show results for binary outcomes - relative risk of
losing 5% or more of body weight over a 12 month period; i.e. the
proportion of treated individuals who met this criterion divided by the
proportion of controls. In 38 trials of behavioural interventions, the
mean RR was 1.94 [95% CI, 1.70 to 2.22]. For 31 pharmaeutical
interventions, the effect varied with the specific medication, with RR
ranging from 1.18 to 3.86. Only two pharmaceutical comparisons had RR in
excess of 3.0. By contrast, for five trials of body contouring or
cellulite reduction from Erchonia, the RRs ranged from 3.6 to 18.0.
Now, it is important to note that this is not comparing like with like:
the people in the Erchonia trials were typically not clinically obese:
they were mostly women seeking cosmetic improvements to their
appearance. So you could, and I am sure many would, argue it's an
unfair comparison. If anyone knows of another literature that might
provide a better benchmark, please let me know. The point is that the
effect sizes reported by Erchonia are enormous relative to the kinds of
effects typically seen with other treatments focused on weight
reduction.
If we look more generally at the other results
obtained with low-level lasers, we can compare them to an overview of
effectiveness of common medications (Leucht et al, 2015).
These authors presented results from a huge review of different
therapies, with effect sizes represented as standardized mean
differences (SMD - familiar to psychologists as Cohen's d). I converted
Erchonia results into this metric*, and found that across all the
studies of pain relief shown in Figure 1, the average SMD was 1.30, with a range from 0.87 to
1.77. This contrasts with Leucht et al's estimated effect size of 1.06
for oxycodone plus paracetamol, and 0.83 for Sumatriptan for migraine.
So if we are to believe the results, they indicate that the effect of
Erchonia low-level lasers is as good or better than the most effective
pharmaceutical medications that we have for pain relief or weight loss.
I'm afraid I remain highly sceptical.
I would not have dreamed of looking at Erchonia's track record if it were not for their impossibly good results in the Leisman et al autism trial that I discussed in the previous blogpost. When I looked in more detail, I was reminded of the kinds of claims made for alternative treatments for children's learning difficulties, where parents are drawn in with slick websites promising scientifically proven interventions, and glowing testimonials from satisfied customers. Back in 2012 I blogged about how to evaluate "neuroscientific" interventions for dyslexia. Most of the points I made there apply to the world of "photomodulation" therapies, including the need to be wary when a provider claims that a single method is effective for a whole host of different conditions.
Erchonia products are sold worldwide and seem popular with alternative
health practitioners. For instance, in Stockport, Manchester, you can
attend a chiropractic clinic where Zerona laser treatment will remove "stubborn body fat". In London there is a podiatry centre that reassures you: "There
are numerous papers which show that cold laser affects the activity of
cells and chemicals within the cell. It has been shown that cold laser
can encourage the formation of stem cells which are key building blocks
in tissue reparation. It also affects chemicals such as cytochrome c and
causes a cascade of reactions which stimulates the healing. There is
much research to show that cold laser affects healing and there are now
several very good class 1 studies to show that laser can be effective."
But when I looked for details of these "very good class 1 studies" they
were nowhere to be found. In particular, it was hard to find research by
scientists without vested interests in the technology.
Of all the RCTs that I found, there were just two that were conducted at reputable universities. One of them, on hearing loss (NCT01820416) was conducted at the University of Iowa, but terminated prematurely because intermediate analysis showed no clinically or statistically significant effects (Goodman et al., 2013). This contrasts sharply with NCT00787189, which had the dramatic results reported in Figure 1 (not, as far as I know, published outside of clinicaltrials.gov). The other university-based study was the autism study based in Boston described in my previous post: again, with unpublished, unimpressive results posted on clinicaltrials.gov.
This suggests it is important when evaluating novel therapies to have results from studies that are independent of those promoting the therapy. But, sadly, this is easier to recommend than to achieve. Running a trial takes a lot of time and effort: why would anyone do this if they thought it likely that the intervention would not work and the postulated mechanism of action was unproven? There would be a strong risk that you'd end up putting in effort that would end in a null result, which would be hard to publish. And you'd be unlikely to convince those who believed in the therapy - they would no doubt say you had the wrong wavelength of light, or insufficient duration of therapy, and so on.
I suspect the response by those who believe in the power of low-level lasers will be that I am demonstrating prejudice, in my reluctance to accept the evidence that they provide of dramatic benefits. But, quite simply, if low-level laser treatment was so remarkably effective in melting fat and decreasing pain, surely it would have quickly been publicised through word of mouth from satisfied customers. Many of us are willing to subject our bodies to all kinds of punishments in a quest to be thin and/or pain-free. If this could be done simply and efficiently without the need for drugs, wouldn't this method have taken over the world?
*Summary files (Erchonia_proportions4.csv) and script (Erchonia_proportions_for_blog.R) are on Github, here.